LINKHIVE Ships 500 Sets of 1B TGG Push Pull Circular Connectors to India
Overview of LINKHIVE Push Pull Circular Connector Shipment
In October 2025, Dongguan Linkhive Connectors Co., Ltd. successfully completed the shipment of 500 sets of 1B TGG push pull circular connectors to a leading medical-device manufacturer in India.
This shipment marks another milestone in the company’s commitment to providing high-quality medical connectors and reliable global logistics solutions.
Each connector was manufactured, assembled, and tested according to ISO 13485 Medical Device Quality System standards.
The 1B TGG series represents one of LINKHIVE’s flagship products—renowned for its miniature structure, high mating accuracy, and stable electrical performance—making it ideal for medical equipment, patient monitoring systems, and diagnostic instruments.
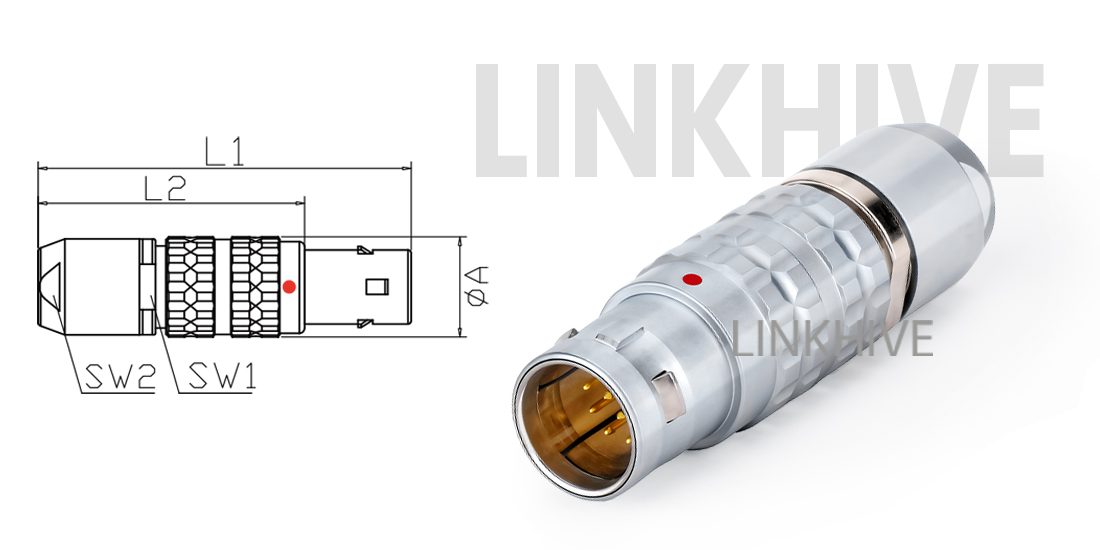
LINKHIVE Push Pull Connector Engineering and Design
The LINKHIVE 1B TGG push pull connector is designed to provide a secure connection in compact medical systems.
It features a self-locking push-pull latch that ensures quick engagement and automatic retention.
The connector unmating operation can be performed safely with a single pull, preventing accidental disconnection during clinical use.
Key technical highlights include:
Precision machining: All shells are CNC-turned with ± 0.01 mm tolerance.
Material selection: Brass with nickel or chrome plating for corrosion resistance and aesthetic finish.
Contact performance: Gold-plated pins for low contact resistance (< 5 mΩ).
Dielectric strength: Rated at 1500 V rms.
Mating cycles: > 5000 operations without mechanical degradation.
Cable diameter range: 4.0 – 6.5 mm.
Each 1B circular connector provides EMC shielding, 360° ground connection, and optional IP66 or IP68 waterproof upgrades for applications requiring frequent sterilization or fluid exposure.

Quality Assurance for Medical-Grade Circular Connectors
To ensure long-term reliability of every LINKHIVE medical connector, the company applies a comprehensive multi-step inspection process.
Every production batch of 1B TGG push pull connectors undergoes:
Material Verification – Raw brass and insulator materials analyzed by XRF spectrometry to guarantee RoHS compliance.
Dimensional Inspection – All shells and inserts checked with digital micrometers and go/no-go gauges.
Surface Treatment Validation – Plating thickness controlled to > 5 μm for nickel and > 0.3 μm for chrome.
Contact Resistance Testing – Random sampling ensures stable electrical conductivity across 100 units.
Mating Force Verification – Insertion and withdrawal forces maintained between 40–80 N.
Final Assembly and Packaging – Each connector individually sealed in anti-static bags for export.
Through these procedures, LINKHIVE guarantees that every push pull circular connector maintains consistent quality across all shipments.
Technical Specifications of the 1B TGG Medical Push Pull Connector
| Parameter | Specification |
|---|---|
| Series | LINKHIVE 1B TGG Push Pull Circular Connector |
| Shell Material | Brass with Nickel or Chrome Plating |
| Insulator | PPSU / PEEK Medical-Grade Plastic |
| Contact Material | Copper Alloy + Gold Plated |
| Pin Configuration | 6 Contacts |
| Rated Voltage | ≤ 1000 V DC |
| Rated Current | ≤ 10 A per Contact |
| Waterproof Level | IP66 / IP68 (optional) |
| Temperature Range | –55 °C to +200 °C |
| Mating Cycles | > 5000 Cycles |
| Shielding Performance | > 65 dB @ 10 MHz |
| Cable OD Compatibility | 4 – 6.5 mm |
This configuration provides a balance between miniaturization, electrical stability, and ease of maintenance—critical features for portable and sterilizable medical devices.
Quality Control Before Shipment of LINKHIVE Push Pull Connectors
Every batch of LINKHIVE circular connectors destined for India underwent both electrical and mechanical validation:
Insulation Resistance Test: ≥ 5000 MΩ @ 500 VDC.
Salt-Spray Test: > 48 hours with no corrosion visible.
Vibration Resistance: Conforms to IEC 60512-6.
Pull-Out Strength: > 90 N axial retention.
Temperature Cycle: –55 °C to +125 °C (10 cycles).
The packaging process follows medical-grade cleanliness protocols.
Connectors are individually bagged in anti-static PE sleeves, packed in foam-lined boxes, and labeled with serial and batch codes for full traceability.

Shipment to India — Ensuring Reliable Delivery of Medical Connectors
LINKHIVE cooperated with 4PX Logistics (S1185 Economic Line) to ensure stable and cost-effective international delivery.
The shipment, totaling 500 sets of 1B TGG connectors, was air-freighted from Shenzhen to Mumbai and then distributed to the customer’s assembly facility in Pune.
Key advantages of this logistics solution:
Reduced transit cost by approx. 60 % compared to DHL Express.
Average delivery time of 10–12 working days.
Continuous tracking and real-time status updates.
The packaging weight of the 500-set shipment was 28 kg, with three layers of impact protection to guarantee safe arrival.
LINKHIVE Push Pull Connector Applications in the Indian Medical Industry
India’s medical device sector is expanding rapidly, especially in diagnostic equipment, patient monitoring, and imaging systems.
The LINKHIVE 1B TGG push pull connectors serve a vital role in these applications thanks to their compact form and reliable connection.
1. Patient Monitoring Systems
Connect ECG and SpO₂ sensors to control modules.
Withstand frequent connection cycles and sterilization.
Provide stable signal transmission with low EMI.
2. Diagnostic Imaging Equipment
Interface modules in ultrasound and endoscopic devices.
Small diameter and shielded design reduce noise.
3. Surgical Instruments
Used in handheld and portable surgical tools requiring lightweight cables.
High-temperature resistance supports autoclave sterilization up to 134 °C.
4. Hospital Data and Sensor Integration
Connects sensors to networked systems in modern hospitals.
Reliable plug-and-play function reduces setup time.
Customer Feedback and Project Continuation
The Indian customer expressed strong satisfaction after testing LINKHIVE samples received during the CMEF Guangzhou exhibition.
Following evaluation of fit and signal integrity, they placed a purchase order for 500 sets of 1B TGG connectors.
Feedback summary:
“The mechanical tolerance and locking structure are perfectly compatible with our existing system.”
“The contact quality and surface finish are equivalent to European brands but at a much more competitive price.”
LINKHIVE has since begun discussions for a second-phase order involving custom blue TGG plugs to match the customer’s corporate color scheme.
Why Indian Customers Choose LINKHIVE Push Pull Connectors
The Indian market values not only product performance but also after-sales support and logistics efficiency.
LINKHIVE meets these expectations through:
Comprehensive Series Coverage – From miniature 0B connectors to IP68 K Series.
Customization Support – Color, engraving, and tailored over-molding.
Fast Sample Service – Delivery within 5 days for standard types.
Competitive Pricing – Cost-effective alternative to LEMO and ODU brands.
Stable Supply Chain – Warehouse stock for regular medical customers.
With continuous technical collaboration, LINKHIVE has established trusted partnerships with medical device manufacturers across Bangalore, Chennai, and Pune.
Comparison of LINKHIVE Push Pull Connector Series
| Series | Shell Material | Sealing Level | Applications | Compatibility |
|---|---|---|---|---|
| 1B Series Push Pull Connector | Brass Chrome | IP50 | Medical equipment, instruments | LEMO B / ODU Mini-Snap L |
| K Series Waterproof Connector | Stainless Steel | IP68 | Outdoor medical devices, sterilization | LEMO K / ODU AMC |
| S Series Circular Connector | Brass Nickel | IP50 | Industrial control systems | LEMO S Series |
| P Series Medical Connector | PPSU Plastic | IP66 | Autoclavable medical equipment | LEMO REDEL P |
| Fluidic Series Hybrid Connector | Brass / SS | IP68 | Pneumatic and gas flow medical systems | LEMO Fluidic / ODU Mediline |
Quality Commitment and Continuous Improvement
Every LINKHIVE connector is the result of precision engineering and decades of connector design experience.
By applying Total Quality Management (TQM) principles, the company ensures that each batch of push pull circular connectors meets the highest international standards.
Recent improvements include:
Upgraded insulator material to medical-grade PEEK for high-temperature resistance.
Enhanced keying depth on 1B and 2C series to prevent rotation and mis-mating.
Double O-ring design for better waterproof performance on K series.
Reinforced strain-relief back nut for repeated bending cycles in mobile devices.
Sustainability and Environmental Responsibility
LINKHIVE connectors are produced under eco-friendly manufacturing standards:
RoHS compliant, no lead or cadmium plating.
ISO 14001 environmental management system certified.
Recyclable metal scrap and solvent collection during machining.
Use of energy-efficient CNC equipment and LED lighting in workshops.
This approach aligns with the sustainability requirements of Indian medical device OEMs and global partners.
Global Accessibility and Support
With customers in Europe, Asia, and the Americas, LINKHIVE Connectors provides rapid response technical support and sample shipment services worldwide.
Indian clients benefit from regional warehouses and customer service teams that offer:
Local engineering advice for connector selection.
Quick replacement and re-supply of critical parts.
OEM / ODM design support for custom connectors and cable assemblies.
These advantages make LINKHIVE a long-term strategic supplier for medical device manufacturers across India.
Conclusion — Delivering High-Quality Medical Push Pull Circular Connectors
The successful shipment of 500 sets of 1B TGG push pull circular connectors to India demonstrates LINKHIVE’s growing presence in the medical equipment market.
With advanced engineering, reliable quality control, and cost-effective logistics, LINKHIVE continues to be recognized as a trusted manufacturer of high-quality connectors for the global medical industry.
LINKHIVE CONNECTORS CO., LTD.
Your Reliable Partner for Medical-Grade Push Pull Circular Connectors.


